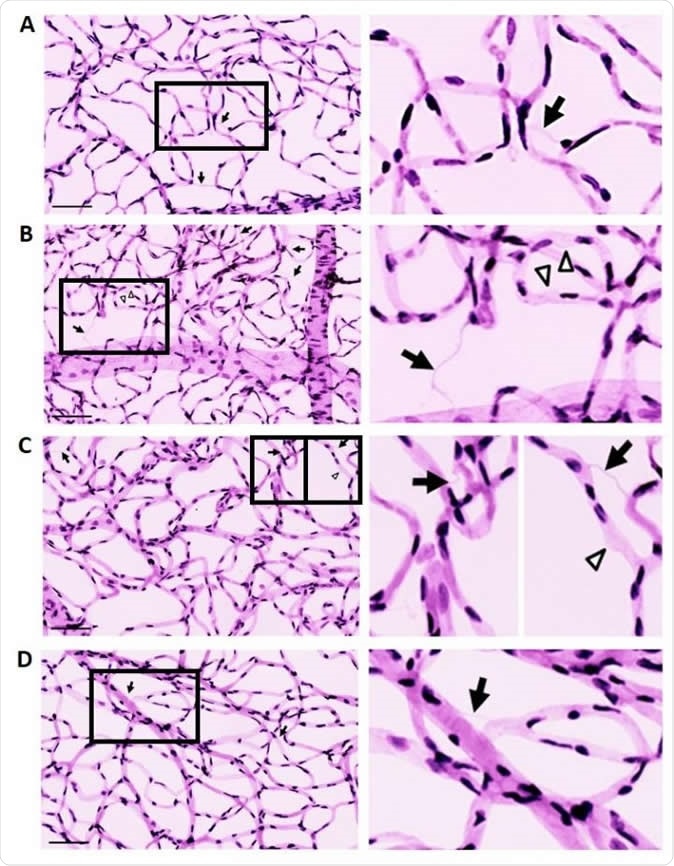
Effect of recombinant lysyl oxidase propeptide (rLOX-PP) on the development of acellular capillaries (ACs) and pericyte loss (PL) in rat retinas. A-D: Representative retinal trypsin digest images showing retinal vascular networks of a control rat (A), diabetic (DM) rat (B), rat intravitreally injected with rLOX-PP (C), and rat intravitreally injected with phosphate-buffered saline (PBS) (D). LOX-PP administration promoted the development of ACs (arrows) and PL (arrowheads) associated with diabetic retinopathy. Enlarged images correspond to boxed areas (Right panels of A, B, C, D). Scale bar = 100 μm.
The study was reported online in The American Journal of Pathology on September 16, 2019.
An enzyme called lysyl oxidase (LOX) is a contributor to cell loss. Prior research indicates that its pro-enzyme form LOX-PP is also involved in triggering the process of programmed cell death, or apoptosis, in unhealthy tissue. Several studies have suggested its contribution to apoptosis by suppressing the AKT pathway by inhibiting the addition of an activating phosphorus group. This is a pathway necessary for cell survival.
The LOX enzyme is involved in strengthening collagen and elastin networks by crosslinking them to provide structural stability in the extracellular matrix, which supports all tissues. However, the role of LOX-PP is not clear, though it has been suggested that it keeps LOX inactive, requires the addition of glucose for the optimal activation of the LOX enzyme, and suppresses several cellular pathways.
This motivated the current study on the effects of LOX-PP (mediated by high blood glucose or diabetes) on the retinal blood vessels.
Diabetic retinopathy
High glucose and diabetes increases LOX-PP levels and cell death
In this experiment, the researchers exposed rat cells from the retinal endothelium to a recombinant form of LOX-PP (rLOX-PP) and to a medium containing high glucose levels. They found that high glucose increases the production of LOX-PP while reducing the activity of the AKT pathway. In the other setup, the cells which had been exposed to rLOX-PP also showed higher levels of cell death, again indicating lower AKT activation.These cells also showed significantly higher levels of the cleaved protein caspase-3 (a marker for the occurrence of apoptosis) when grown in a high-glucose medium. The number of apoptotic cells was significantly increased as well, both in the cells grown in a high-glucose medium and those which were first grown in normal medium but then exposed to rLOX-PP for 24 hours. In contrast, exposure to mannitol after growth in normal medium had no significant effect.
When the retinas of diabetic rats were examined, AKT phosphorylation, and therefore, activation, was significantly less. This was mimicked by injecting rLOX-PP into the vitreal space of normal rats. In both cases, the number of apoptotic cells was significantly higher while pericyte loss was reduced. This is characteristic of DR. Thus this proenzyme suppresses the AKT pathway.
The results also agree with previous studies which show that an increase in LOX in the presence of diabetes or a high-glucose medium thickens and stiffens the vascular basement membrane of the retinal vessels. However, higher levels of LOX are by definition linked to increased concentrations of LOX-PP, it is possible that the loss of retinal vascular cells is in part due to, or accompanied by changes in the basement membrane of the blood vessels. This was confirmed by the similarity of the changes seen in the retinal cells after being exposed directly to rLOX-PP.
Conclusion
Lead researcher Sayon Roy says, “This report shows novel functionality of LOX-PP in mediating cell death under high glucose condition in retinal endothelial cells as well as in diabetic animals.”In effect, high glucose levels increase the levels of LOX-PP which in turn causes retinal endothelial cells to undergo apoptosis at higher rates. This could therefore be the mechanism that is active in diabetic retinopathy, via the suppression of the AKT pathway.
The presence of this link will make it necessary to explore deeper into the specific manner in which this proenzyme produces apoptosis – whether by inhibition of fibroblast growth factor 2 signaling. Meanwhile, the current study does offer hope for the emergence of a therapy for diabetic retinopathy by targeting LOX-PP.
it is also noteworthy that LOX-PP also makes cancer cells more sensitive to the effects of the chemotherapeutic drug doxorubicin, which causes apoptosis. But scientists don’t know whether this effect is the same as that operating in the current study. However, they do know that LOX-PP makes cells more prone to DNA break down or splitting by disabling the normal DNA repair proteins. It is also known that high glucose levels also cause DNA breakdown. Thus more research is needed to understand whether LOX-PP is involved in increasing the strength of all these effects produced by high glucose levels.
Journal reference:
Effects of high glucose–induced lysyl oxidase propeptide on retinal endothelial cell survival iimplications for diabetic retinopathy. Dongjoon Kim, Dayeun Lee, Philip C. Trackman, & Sayon Roy. The American Journal of Pathology. September 16, 2019. DOI: https://doi.org/10.1016/j.ajpath.2019.06.00. https://ajp.amjpathol.org/article/S0002-9440(18)30488-7/fulltext






No comments
Post a Comment